| Issue |
Metall. Res. Technol.
Volume 123, Number 2, 2026
Special Issue on ‘Innovations in Iron and Steelmaking’, edited by Carlo Mapelli and Davide Mombelli
|
|
|---|---|---|
| Article Number | 219 | |
| Number of page(s) | 6 | |
| DOI | https://doi.org/10.1051/metal/2026014 | |
| Published online | 24 February 2026 | |
Original Article
Carbothermic reduction of hot rolling sludge by rice husk char with microwave heating
1
Faculty of Engineering, Kyushu University, 744 Motooka, Nishi-ku, Fukuoka 819-0395, Japan
2
Yamato Kogyo Co., Ltd., 380 Yoshimi, Otsu-ku, Himeji, Hyogo 671-1132, Japan
* e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
1
September
2025
Accepted:
7
January
2026
Abstract
The scale generated in the hot rolling process is a high-grade iron source. Fine scales are separated in a thickener and discharged as sludge in a slurry state, which makes them difficult to use. This study focused on sludge recycling through carbothermic reduction using microwave heating. Microwave heating occurs when an object absorbs microwaves. Compared with conductive heat transfer, microwave heating allows for internal heating, which makes it effective for heating fine powders with low thermal conductivity. To achieve carbon neutrality, the carbothermic reduction behavior was investigated using biomass-derived carbon. For the experiments, industrial sludge, rice husk char as biomass and reagent-grade graphite were used. The sludge had a composition of T. Fe = 64.1 wt.% and FeO = 36.2 wt.%. The biomass was obtained by pyrolyzing rice husks at 500 °C, containing 43.2 wt.% carbon with 47.7% SiO2 as the main impurity. The biomass or graphite and sludge were mixed in a molar ratio the carbon and oxygen content in the sludge matched C/O = 1. Microwave heating was carried out with 2.45 GHz multimode irradiation with magnetron under a nitrogen atmosphere. The total heating time was 20 min, with a maximum output intensity of 1050 W. The samples were heated from room temperature to 1200 °C and then maintained at this temperature. After heating, the formation of metallic iron was confirmed in both samples. During microwave heating, the sample using biomass took longer to stabilize at 1200 °C compared with graphite. It seems due to the SiO2 in the biomass not absorbing microwaves. SEM-EDS analysis of the reduced samples confirmed the presence of metallic iron and residual FeO in both samples. Additionally, in the biomass sample, a large amount of silicate slag containing iron oxides was observed.
Key words: microwave heating / carbothermic reduction / biomass / sludge recycling / rice husk char
© T. Tokunaga et al., Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
In recent years, research and development aimed at achieving sustainable steelmaking has been actively pursued in the steel industry [1]. One such approach is carbon-neutral steel production, which involves the utilization of hydrogen and non-fossil green energy sources [2,3]. In addition, studies have been conducted on the reuse of waste materials from existing processes [4,5] The present study focuses on scale discharged from the hot rolling process. Many steel products such as shape steel, steel plates, and steel bars are industrially manufactured through the hot rolling process. Steel materials are heated in a furnace and then transported to the rolling mill in an atmospheric environment. At that time, because the steel reaches a high temperature, an oxide film (scale) is formed on the surface [6]. The removed scale is separated in a settling tank: small particles float and large particles settle. The settled scale is recovered, and the floated scale becomes a sludge-like substance by adsorbing machine oil mixed into the settling tank, and then it is transferred to the thickener. The sludge is concentrated and discharged by a scraping blade. The discharged sludge contains a lot of moisture, so it is dried in the sun. In 1996, approximately 800,000 tons of sludge [7] were recovered from 107.9 million tons of crude steel production in Japan [8]. Assuming it was generated in the same proportion, based on the crude steel production of 87 million tons in 2023 [9], it is calculated that 690,000 tons of sludge were recovered. Since the scale in the sludge is derived from steel materials, it contains few impurities, and the T.Fe content is as high as 74.4 wt.% in low-carbon rimmed steel scale [10]. Therefore, large-scale is reused as an iron source in the production of sintered ore [11]. On the other hand, sludge with oil and moisture attached to the scale is difficult to reuse. The reasons include the difficulty of powder compaction due to the moisture contained in the sludge, and the possibility of steam explosions occurring inside the furnace when attempting to reuse it using an electric furnace. As a method to enable reuse, removing moisture from sludge by heating can be considered. Furthermore, since the sludge contains carbon originating from machine oil adhering during the descaling and recovery processes, it is expected that reduction reactions will proceed during heating. However, since sludge is a powder with particle size less than 1 mm and has low thermal conductivity [12], conventional external heating requires a long heating time, resulting in large energy loss. Therefore, as an alternative, a method of heating sludge using a technique that does not rely on conductive heat transfer is expected.
The penetration depth of magnetite, which is contained in hot rolling sludge and significantly affects microwave absorption, is 80 micrometers [13]. Therefore, while it cannot completely overcome conductive heat transfer, it serves as a heating method where the influence of conductive heat transfer between particles is relatively small. Microwave heating can be used to rapidly raise the temperature in a short time through internal heating. Microwaves are a type of electromagnetic wave. Substances that can be heated by microwaves include dielectrics, magnetic materials, and non-magnetic metal powders. The heating of these substances is caused by electric or magnetic fields. The microwave absorption energy P [W/m3] is expressed by equation (1).
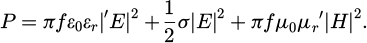 (1)
(1)
Here, E is the electric field (V/m), H is the magnetic field (A/m), σ is the electrical conductivity (S/m), f is the frequency (Hz), ε0 is the vacuum permittivity (F/m), εr" is the imaginary part of the permittivity (-), μ0 is the vacuum permeability (H/m), and μr′ is the imaginary part of the permeability (-) [14]. Equation (1) represents the sum of dielectric loss, resistance-related loss of the material (Joule loss), and magnetic loss, indicating that these three distinct mechanisms contribute to microwave heating [15]. Microwave heating enables selective heating, as the absorption of microwaves varies by material. This selective absorption allows for targeted and energy-efficient reactions [16,17].
When heating sludge with microwaves, it is considered that adding carbon with high microwave absorption efficiency externally can accelerate the rate of temperature rise and promote reduction. L. M. da Silva et al. discussed the carbon thermal reduction of a mixture of iron ore, steelmaking mill scale, and coke fines using microwaves [18]. Our research focuses on the carbon thermal reduction of sludge, which has a smaller particle size than mill scale. By reducing iron oxide in the sludge to metallic iron through the addition of carbon material, it becomes possible to recycle it in a similar manner to scrap. At that time, by using rice husk char (RHC)—currently used as insulation material in steelmaking—as the carbon source to be added, it is possible to reduce actual CO2 emissions. RHC is biomass produced by incomplete combustion and carbonization of rice husks. A huge quantity of rice husk (ca. 3 million tonnes) is produced every year as agricultural waste in Japan [19,20], and it contains approximately 40% fixed carbon. Since it contains a high amount of SiO2 and has a low carbon content, it is considered unsuitable for use as bioethanol or as a fuel [20]. Therefore, the use of RHC as a reducing agent was considered by adding it to the sludge.
This study aimed to clarify the dehydration behavior of sludge during microwave heating and to investigate the moisture removal rate during microwave heating. In addition, the study aimed to clarify the effect of adding RHC during microwave heating of sludge by measuring temperature changes and reflected waves during microwave irradiation to investigate the heating characteristics, and to examine the progress of reduction caused by the carbon contained in the sludge and carbon added externally.
2 Materials and methods
In this study, sludge recovered from the hot rolling process (supplied by Yamato Steel Co., Ltd.), RHC (supplied by Yamato Steel Co., Ltd.), and graphite reagent (particle size ≈ 75 μm, purity 99%, Kishida Chemical Co., Ltd.) were prepared. Sludge contained iron oxide, moisture, and rolling mill oil; it contains carbon. Sludge sieved to <1 mm without drying was used as the wet sample, and the sample dried at 120 °C in an air atmosphere for 24 hours using a muffle furnace and then sieved to less than 1 mm was used as the dry sample. The RHC was preliminarily processed by grinding and sieved to a particle size of <75 μm. The composition of the RHC and dry sample are shown in Table 1. T.C. is an abbreviation for Total Carbon, which includes both the volatile and solid carbon components present in RHC. C.W. is an abbreviation for Combined Water, representing the total water content in RHC.
An energy-dispersive X-ray fluorescence analyzer (Malvern Panalytical, Netherlands) was used for compositional analysis. A dry sample was measured and considering the carbon content in the sludge to achieve a C/O ratio of 1 relative to the oxygen in the sample, charcoal was added to prepare mixed samples. The weights of sludge and graphite in the graphite mixed sample, and the weights of sludge and RHC in RHC mixed samples are shown in Table 2. Since RHC releases a large number of volatile substances, considering safety during the experiment, tests were conducted using 5 g samples. The progress of reduction by microwave heating was investigated for the dry sample, the graphite mixed sample, and RHC mixed sample. The apparatus used in this experiment is a multimode cavity-type microwave furnace (Shikoku Instrumentation Co., Ltd.) with a maximum output power of 1.5 kW at 2.45 GHz. This device sets the target temperature and holding time, detects the temperature of the heating target with a radiant heat meter, and automatically performs appropriate microwave blocking operations and output changes [20]. A detailed schematic diagram of the sample arrangement method is shown in Figure 1. The time-dependent change in the surface temperature of the sample was measured using a two-color thermometer (IR-FAQINN, Chino). To observe the cross-section of the sample after heating, scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDS) were performed for elemental composition analysis. SEM observation was conducted using a low-vacuum high-sensitivity scanning electron microscope (SU3500, Hitachi High-Tech), and elemental analysis was performed using an energy-dispersive X-ray analyzer (Apollo SDD, AMETEK). In Experiment 1, 10.0 g of wet sample was filled into a crucible, N2 was flowed at 1.0 NL/min, and microwave radiation was applied at a maximum output of 800 W. Water removal was considered complete when the sample temperature exceeded 330 °C, which is the lower measurement limit of the two-color thermometer. In Experiments 2–4, dry sample, graphite mixed sample, and RHC mixed samples were filled into crucibles, N2 was flown at 1 NL/min, and microwaves were irradiated with a maximum output of 1050 W and a target temperature of 1200 °C with the aim of melting metallic iron and separating it from oxides. The microwave output was regulated by PID control to maintain the set temperature.
Chemical composition (mass%) of sludge and rice husk char.
The weights of sludge and graphite in the graphite-mixed sample, and RHC in the rice husk-mixed sample.
3 Results and discussion
In experiment 1, it was conducted using wet samples to compare moisture removal by microwave heating and conventional heating methods. When the sample was heated to 120 °C in a muffle furnace, the weight change stopped after 22 hours. Based on the weight reduction of the sample, the moisture content of the sludge was 17.7%. The weight change during dehydration by microwave heating was 13.2% at 111 s, suggesting that 74.6% of the total moisture was removed.
The weight change rate during dehydration by the muffle furnace reached 13.0% after 250 min had elapsed since placing the sample in the 120 °C furnace. This suggests that microwave heating can heat powders and remove moisture in a shorter time [21]. Next, the heating characteristics of moisture-containing sludge were examined based on the microwave absorption ratio. The microwave absorption ratio was derived using equation (2) shown below.
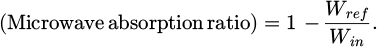 (2)
(2)
Here, Win is the incident microwave energy (J/s), and Wref is the reflected microwave energy (J/s). Figure 2 shows the microwave absorption ratio during microwave heating of the wet sample. The microwave absorption ratio began to decrease 50 s after heating started, and then increased sharply just before the sample temperature reached 750 °C. At temperatures below 330 °C, the substance that functions as a good absorber of microwaves in the sludge is water molecules, which absorb microwaves through dielectric loss due to their polarity [22]. This suggests that the decrease in microwave absorption ratio due to the reduction in water molecules indicates that moisture removal began 50 s after heating started. Furthermore, the increase in microwave absorption ratio just before irradiation cessation is attributed to the properties of hematite, which becomes a good microwave absorber due to the increase in dielectric constant and conductivity around 400 °C [23].
In experiment 2, 10.0 g of dried sample was heated with microwaves for 5 min to investigate the heating characteristics of the sludge. The variations of sample temperature and microwave absorption ratio during heating are shown in Figure 3. The sample temperature exceeded 330 °C 42 s after heating, then began to decline, reaching 1037 °C at 300 s. During heating, temperature measurement was paused for 5 s at the 100 s mark to check the sample condition. The microwave absorption ratio reached a maximum of 0.78 at 42 s, when the temperature increase was confirmed, and gradually decreased to 0.51 at 300 s. From these results, it is considered that temperature and microwave absorption ratio showed similar trends, and that the target temperature could not be maintained because the proportion of microwave energy absorbed by the sludge decreased over time.
The results of XRD analysis performed on the samples before and after heating are shown in Figure 4. Based on XRD analysis, it is considered that microwave heating of the dried sample caused the carbon contained in the sludge to act as a reducing agent, reducing hematite and magnetite to wüstite. These findings suggest that the failure to maintain the target temperature in the latter part of heating was due to the reduction of hematite and magnetite, which decreased the proportion of good microwave absorbers in the sample and led to a decline in microwave absorption ratio [21].
In experiment 3 and 4, the graphite mixed sample and the RHC mixed sample were heated with microwaves for 20 min. Figure 5 shows the time-dependent changes in temperature and microwave absorption ratio during microwave heating of the mixed samples. In the mixed samples, the temperature was maintained at 1200 °C and the microwave absorption ratio was maintained at 0.8 or higher. On the other hand, when comparing these results, the graphite mixed sample reached 330 °C, and the time required to stabilize at 1200 °C was shorter than that required for the RHC mixed sample. Since SiO2 has a property of transmitting microwaves [24], it is presumed that RHC, which contains high SiO2 content, is less susceptible to microwave heating than graphite. From these results, it was found that, while the SiO2 content in RHC tends to prolong the time required for temperature increase, RHC can be expected to enhance microwave absorption similarly to graphite.
Cross-sectional observations were made using SEM-EDS on the heated samples. Figure 6a shows SEM images of graphite mixed samples, and Figure 6b shows SEM images of RHC mixed samples. Both samples showed that metallic iron had been generated, as there were areas where Fe and O peaks did not overlap in
either case. As another phase, the graphite mixed sample contained what is thought to be FeO and SiO2, while the RHC mixed sample contained a phase in which Fe, Si, and O coexisted. This is due to the increased proportion of SiO2 resulting from the addition of RHC. These findings indicate that, by mixing RHC and applying microwave heating, the carbon in the RHC acts as a reducing agent, enabling partial reduction of iron oxides in the sludge to metallic iron.
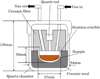 |
Fig. 1 Schematic diagram of the sample arrangement in microwave furnace. |
 |
Fig. 2 Microwave absorption ratios of experimental 1 (wet sample). |
 |
Fig. 3 Temperature profiles and microwave absorption ratios of experimental 2 (dry sample). |
 |
Fig. 4 XRD analysis of sample before and after microwave heating experiment 2 (dry samples). |
 |
Fig. 5 Variations of temperature and microwave absorption ratios of experimental 3 (graphite mixed sample) and experimental 4 (RHC mixed sample). |
 |
Fig. 6 SEM-EDS (15.0 kV × 6.50 k) images of: (a) experimental 3 (graphite mixed sample); (b) experimental 4 (RHC mixed sample). |
4 Conclusion
This study investigated the dehydration and reduction behavior of the sludge during microwave heating and the effect of RHC addition. The main findings are as follows:
Microwave heating of wet sludge raised the surface temperature above 330 °C, resulting in 74.6% moisture removal within 111 s.
Heating the dry sludge at 1200 °C for 300 s reduced hematite and magnetite to wüstite. The temperature and microwave absorption peaked at 42 s and subsequently declined, owing to the consumption of hematite, which is a strong microwave absorber.
In graphite-mixed samples heated at 1200 °C for 20 min, temperature and absorption decreases were suppressed, and metallic iron was partially formed. This is attributed to graphite enhancing microwave absorption.
RHC addition similarly maintained high absorption and promoted partial reduction to metallic iron, demonstrating that the carbon in RHC functions effectively both as a microwave absorber and a reducing agent.
Funding
This research was funded by Yamato Kogyo Co., Ltd., under a collaborative research agreement.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author contribution statement
The research was conceived and supervised by Ko-ichiro Ohno.
The joint research project was initiated and coordinated in collaboration with Kotaro Tanaka, who also contributed to technical discussions throughout the study.
Tatsuya Kon developed the experimental methodology and assisted with data analysis.
Tsubasa Tokunaga performed the experiments and prepared the initial manuscript draft.
The manuscript was reviewed and edited by Ko-ichiro Ohno and Tatsuya Kon.
All authors have read and approved the final version of the manuscript.
References
- T. Ariyama, Perspectives on the promising pathways to zero carbon emissions in the steel industry toward 2050, ISIJ Int. 65, 165 (2025) [Google Scholar]
- F. Mauret, J. Ji, P. Kinzel et al., Ammonia as a green energy carrier to lower blast furnace CO2 emissions, ISIJ Int. 65, 601 (2025) [Google Scholar]
- K. Momono, J. Ishii, S. Hosohara et al., Development of innovative gasification process of used plastic by using fluidized bed, ISIJ Int. 64, 954 (2024) [Google Scholar]
- O. Vitikka, M. Iljana, A. Heikkilä et al., Effect of biocarbon addition on metallurgical properties of mill scale-based auger pressing briquettes, ISIJ Int. 64, 964 (2024) [Google Scholar]
- O. Vitikka, M. Iljana, A. Heikkilä et al., Suitability of auger pressing briquettes for blast furnace use based on laboratory tests, Minerals. 12, 868 (2022) [Google Scholar]
- M. Krzyzanowski, J.H. Beynon, D.C.J. Farrugia, Oxide scale behavior in high temperature metal processing, Wiley-VCH, Weinheim, 2010 [Google Scholar]
- T. Uchiyama, Y. Hara, K. Takeda et al., Development of recovery technology of valuable metal contained in industrial sludge by smelting reduction furnace with two-stage tuyeres, Tetsu-to-Hagané. 86, 56 (2003) [Google Scholar]
- T. Fujiwara, Production and technology of iron and steel in Japan during 1996, ISIJ Int. 37, 305 (1997) [Google Scholar]
- The technical society of the ISIJ, production and technology of iron and steel in Japan during 2023, ISIJ Int. 64, i (2024) [Google Scholar]
- I. Endou, Y. Morioka, S. Itoh, Reduction of packed milled-scale bed by solid reducing agents, J. Jpn. Inst. Met. 40, 105 (1976) [Google Scholar]
- M. Kaimoto, R. Shiozaki, K. Matsuyama et al., Improving of productivity on sinter operation by preparatory granulation method in high blending ratio of mill scale, Tetsu-to-Hagané. 103, 36 (2017) [Google Scholar]
- A. Yoshiaki, J. Soc. Powder Technol. Jpn. 22, 161 (1985) [Google Scholar]
- M. Hayashi, Y. Yokoyama, K. Nagata, Effect of particle size and relative density on powdery Fe3O4 microwave heating, J. Microwave Power Electromagn. Energy. 44, 198 (2010) [Google Scholar]
- A. Amini, K. Ohno, T. Maeda et al., Effect of the ratio of magnetite particle size to microwave penetration depth on reduction reaction behaviour by H2, Sci. Rep. 8, 15023 (2018) [Google Scholar]
- M. Tanaka, H. Kono, K. Maruyama, Selective heating mechanism of magnetic metal oxides by a microwave magnetic field, Phys. Rev. B. 79, 104420 (2009) [Google Scholar]
- K.O., Mechanism and applications of microwave heating, J. Jpn. Soc. Colour Mater. 44, 27 (1971) [Google Scholar]
- M. Ignatenko, M. Tanaka, M. Sato, Absorption of microwave energy by a spherical nonmagnetic metal particle, J. Appl. Phys. 48, 067001 (2009) [Google Scholar]
- L.M. da Silva, T.F. Eugenio, G.A. de Medeiros et al., Evaluation of the use of microwave energy on the reduction of iron ore and steelmaking mill scale composite self-reducing mixtures, Mater. Res. 25, 1 (2022) [Google Scholar]
- K. Suzuki, S. Suzuki, M. Fujita, Industrial applications of rice husk, Mokuzai Kogyo. 57, 192 (2002) [Google Scholar]
- S. Kumagai, K. Ikawa, K. Takeda, Ammonia gas adsorption by carbonized rice husk, Int. J. Soc. Mater. Eng. Resour. 13, 92 (2006) [Google Scholar]
- P. Lunkenheimer, S. Emmert, A. Loidl, R. Gulich et al., Electromagnetic -radiation absorption by water, Phys. Rev. E. 96, 062607 (2017) [Google Scholar]
- M. Al-Ali, K.I. Salih, A. Alsamarrae, Microwave heating temperatures and pharmaceutical powder characteristics, Mater. Today. 20, 583 (2020) [Google Scholar]
- Z. Peng, J.Y. Hwang, C.L. Park et al., Microwave permittivity, permeability, and absorption capability of ferric oxide, ISIJ Int. 52, 1535 (2012) [Google Scholar]
- H. Fukushima, Electromagnetic properties of silicates, Chem. Educ. 54, 450 (2006) [Google Scholar]
Cite this article as: Tsubasa Tokunaga, Tatsuya Kon, Kotaro Tanaka, and Ko-ichiro Ohno, Carbothermic reduction of hot rolling sludge by rice husk char with microwave heating. 123, 219 (2026) https://doi.org/10.1051/metal/2026014
All Tables
The weights of sludge and graphite in the graphite-mixed sample, and RHC in the rice husk-mixed sample.
All Figures
 |
Fig. 1 Schematic diagram of the sample arrangement in microwave furnace. |
| In the text | |
 |
Fig. 2 Microwave absorption ratios of experimental 1 (wet sample). |
| In the text | |
 |
Fig. 3 Temperature profiles and microwave absorption ratios of experimental 2 (dry sample). |
| In the text | |
 |
Fig. 4 XRD analysis of sample before and after microwave heating experiment 2 (dry samples). |
| In the text | |
 |
Fig. 5 Variations of temperature and microwave absorption ratios of experimental 3 (graphite mixed sample) and experimental 4 (RHC mixed sample). |
| In the text | |
 |
Fig. 6 SEM-EDS (15.0 kV × 6.50 k) images of: (a) experimental 3 (graphite mixed sample); (b) experimental 4 (RHC mixed sample). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


