| Issue |
Metall. Res. Technol.
Volume 123, Number 2, 2026
Special Issue on ‘Innovations in Iron and Steelmaking’, edited by Carlo Mapelli and Davide Mombelli
|
|
|---|---|---|
| Article Number | 209 | |
| Number of page(s) | 10 | |
| DOI | https://doi.org/10.1051/metal/2025138 | |
| Published online | 05 February 2026 | |
Original Article
Comparison of methods for modification of non-metallic inclusions using alkali elements on a laboratory scale
1
Christian Doppler Laboratory for Inclusion Metallurgy in Advanced Steelmaking, Technical University of Leoben, Franz Josef-Straße 18, 8700 Leoben, Austria
2
Voestalpine Stahl Donawitz GmbH, Kerpelystraße 199, 8700 Leoben, Austria
* e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
22
September
2025
Accepted:
8
December
2025
Abstract
The cleanness of modern steel is of utmost importance as non-metallic inclusions (NMIs) can have detrimental effects on the mechanical properties of the finished product. Hard and non-deformable inclusions can act as crack initiation sites, reducing fatigue strength, notched impact toughness, and formability. Even small populations of such NMIs can impair the properties of cleanness-sensitive grades and may also cause operational problems such as nozzle clogging during production. Adapted deoxidation and refining practices like Silicon- (Si)/ Manganese- (Mn) deoxidation can reduce the amount of Aluminium- (Al) rich NMIs, and slag refining treatments of the melt during secondary metallurgical operations can aid in improving steel cleanness. Nonetheless, complete elimination of NMIs is not feasible, highlighting the need for strategies that directly improve the quality of remaining inclusions. This study investigates the potential of alkali elements, specifically sodium (Na) and potassium (K), to chemically modify inclusions during secondary refining. Experiments were performed using alkali oxide-containing slags and combined slag-carbonate treatments in two steel grades. Manual and automated SEM/EDS analysis were used to evaluate both indirect and direct modification effects. Results indicate that treatment using alkali oxide-containing slags can achieve indirect modification of the inclusion landscape, reflected in a reduced relative amount of sulfides. Above all, it could be demonstrated that a combined slag-carbonate treatment is necessary to enable direct incorporation of Na and K into NMIs. These alkali elements predominantly modify Al-containing phases, including alumina-rich phases within multiphase particles. Thermodynamic calculations using FactSage 8.3 predict a decreased liquidus temperature of Al2O3 in the presence of Na2O or K2O. Future work will show the impact of this modification on the mechanical properties of the inclusions and the steel.
Key words: non-metallic inclusions / alkali elements / inclusion modification / steel cleanness / inclusion control / secondary refining
© N. Preisser et al., Published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Non-metallic inclusions (NMIs) are an ever-present part of all grades and qualities of steel. Various particles and impurities found in the steel's matrix can be labeled as NMIs. Exogenous NMIs comprise all particles brought into the melt from the outside, like entrapped slags, eroded particles from refractory material, or other debris that comes into contact with the melt. Particles formed within the melt due to thermodynamic reactions are called endogenous NMIs and comprise deoxidation products alongside all other non-metallic phases formed during production in the melt. As exogenous NMIs are typically much larger, the removal via flotation is usually no problem in modern steel production. Endogenous particles are often much smaller and not as easy to remove, especially as they can be continuously formed during the production of steel [1]. While great efforts are taken to minimize the amount of NMIs in modern cleanness-sensitive steel grades, it is virtually impossible to remove all and every NMI from the melt.
As NMIs usually show different mechanical properties than the surrounding steel matrix, they can seriously affect the steel products’ properties, including fatigue strength, toughness, or formability [2–5]. This is due to the formation of voids between hard and non-deformable NMIs, such as aluminum oxides, and the much softer steel matrix, which in turn can act as initiation sites for crack formation [6–10]. To counteract this, efforts have been made by various researchers to modify the inclusion landscape in cleanness-sensitive steel grades. By additions of elements like lithium, sodium, potassium, or even rubidium to the slag or the melt NMIs are sought to be removed [3,11–14] or modified to alter their chemical composition [4,15,16].
Jiang et al. [14] found that additions of up to 7.5 wt.% of K2O to ladle furnace refining slag lead to reduced size and number of inclusions in samples of saw wire steel. Remaining inclusions were found to mainly concentrate in low melting zones of SiO2-MnO-Al2O3 or SiO2-CaO-Al2O3 systems. Li et al. [12] found the same effect for additions of K2O to refining slags, whereas Li2O led to contrasting results where inclusion size and amount rose after refining. Chen et al. [3,17] reported that additions of Na2O to refining slags led to increased amounts and size of NMIs but found that Rb2O could decrease the size and number of inclusions in samples of saw wire steels. Regarding direct additions of Na2CO3 to the melt, Chen et al. [18] found that inclusions could be directly modified to contain amounts of Na2O. Also, additions of NaF to the melt were found to be capable of directly modifying NMIs in saw wire steel samples [19]. Zhao et al. [20] reported direct modification of NMIs in high-carbon steels by additions of Na2CO3 and K2CO3 to the melt.
In this work, impacting the overall inclusion landscape by, for example, lowering the relative amount of oxides is referred to as indirect modification, whereas a change in the chemical composition of NMIs to include added alkali-elements is referred to as direct modification of NMIs. While previous studies have investigated individual methods of impacting the inclusion landscape of different steel grades, this work directly compares different experimental set-ups and procedures for alkali-element treatments using Na- and K-bearing components, such as slag-only, carbonate-only, as well as combined approaches. The aim of this work is to gain a deeper understanding of the effect of adding alkali-elements to the steel and slag phase. Hence, these three methods are systematically evaluated regarding their impact on the indirect and direct modification of NMIs, as well as their practicability.
2 Thermodynamic considerations
For the purpose of planning and managing expectations of the experiments, thermodynamic calculations regarding relevant reactions and key systems have been performed in advance. First and foremost, reactions that take part in the thermal decomposition of the alkali-carbonates are of interest in the course of this work. As calculated with FactSage 8.3 [21] for Li2CO3, the thermal decomposition reaction is exothermic, while for Na2CO3 and K2CO3, this reaction is endothermic at a temperature of 1600 °C. Contrary, the reduction reaction of Li2O to metallic Li with CO is endothermic, while the reduction of Na2O and K2O is exothermic at this temperature. The reaction enthalpies are shown in Table 1. Additionally, the impact of alkali-oxide modification of Al2O3 regarding the liquidus temperature was calculated. With the addition of both Na2O as well as K2O, the liquidus temperature of the system decreases. Up to approximately 10 wt.% of the respective alkali metal oxide, the decrease in liquidus temperature is nearly identical. With higher amounts of Na2O, the liquidus temperature decreases drastically, while further additions of K2O lead only to a slight decrease. Pure alumina shows a liquidus temperature of about 2054 °C, with 24.8 wt.% of Na2O a eutectic composition is reached between Al2O3 and NaAlO2, which melts at temperatures of 1585 °C. A second eutectic composition is reached at 92.57 wt.% of Na2O between NaAlO2 and Na2O with a melting temperature of 1089 °C. For additions of K2O, a eutectic point is found at 30.5 wt.% of K2O between KAlO2 and Al2O3 at temperatures of 1919 °C. Pure KAlO2 melts at 2235 °C, while pure K2O shows the lowest melting temperature in this system at 739 °C.
For a deeper insight into the implications of alkali element additions on sulfidic NMIs the solubility of sulfur in the slag was calculated. Calculations were performed for a medium carbon steel grade, containing 200 ppm of S, alloyed with Mn and Si. This steel was brought into equilibrium with different slag compositions. The desired chemical compositions of those slags are shown in Table 2, where M2O is representative for Na2O and K2O, respectively. Additionally, the composition of slags used for the experimental part of this work as measured by X-Ray Fluoresence Spectroscopy (XRF) are given in parentheses in this table. As Na can not be measured by this technique, it was not possible to give the actual composition of slag N5A5, but the relation of SiO2:CaO:Al2O3 measured is similar to the other two slags. It can be seen, that measured values of SiO2 content are higher than desired contents, whereas measured CaO contents are lower than the desired values. Measured Al2O3 contents in slag REF and K5A5 are slightly higher than the desired values. The measured value of K2O content agrees well with the desired value for slag K5A5.
The mass of slag used in these calculations amounted to 5% of the steels mass. From these calculations the mass of S present in each phase was divided by the initial amount of S in the system, to show the percentage of total amount of S present in each phase at different temperatures. The results of these calculations are shown in Figure 1. In each subplot the colored lines show the relative amount of S present in the liquid phase, the slag phase, as well as the sulfides in equilibrium with alkali oxide-containing slags, whereas the dotted lines show the respective amounts of S in equilibrium with slag REF, without any alkali elements. It can be shown that all investigated alkali elements have a distinct effect on the S-solubility of the system. Especially, the amount of S soluble in the respective slag phases is greatly enhanced in comparison to slag REF.
Not only is the amount of S soluble in the slag phase affected, but also the temperature interval in which S is most stable in the slag phase is expanded. In equilibrium with slag REF S-solubility peaks only for a small temperature interval at about 70% of total S in the slag, whereas with additions of alkali elements to the slag phase the temperature-range of high S-stability in the slag is increased and nearly all the S is present in the slag phase for slags N5A5 and K5A5. The results of these calculations indicate that during slow-cooling from the liquid phase, S can move from the melt to the liquid slag phase. As the steels solidus temperature (1351 °C) lies in the same temperature range of high S-solubility in the slag, S can not migrate back into the steel to form sulfides at lower temperatures. Conversely, rapid cooling or quenching should hinder this migration to the slag phase and facilitate sulfide formation in the steel phase.
Reaction enthalpies of relevant reactions as calculated with FactSage 8.3.
Desired slag compositions and composition as measured by XRF (in parentheses), where M2O stands for Na2O or K2O, respectively.
 |
Fig. 1 Solubility of Sulfur in steel-, slag, and sulfide phase in contact with a) Na2O-containing slag, b) K2O-containing slag. |
3 Development of experimental setup
Multiple experimental methodologies and setups have been developed and tested to determine the capability of modifying NMIs using alkali elements. In the first experiments this should be achieved solely via the slag phase. The second set of experiments was conducted by adding carbonatic alkali elements directly to the melt. Lastly, coupled experiments using both an alkali element-containing slag as well as additions of carbonatic alkali compounds were performed.
3.1 Slag modification experiments
For experiments incorporating the use of slag, slags have been prepared by melting the raw materials, namely Al2O3, SiO2, and CaO, in combination with Na2CO3 or K2CO3, respectively. During heating of these powdered and well-mixed materials, the carbonatic alkali compounds were subject to thermal decomposition of the carbonates to oxides and CO2, as shown in equation (1).
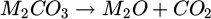 (1)
(1)
Here, M is a placeholder for either Na or K, respectively. Slags were heated using an induction furnace (Indutherm MU 900) to 1600 °C and held for 15 min to ensure complete liquefication and solution of all raw materials as well as complete thermal decomposition of the carbonatic alkali compounds. Due to the volatile nature of the alkali oxides, these were subject to some amount of losses during the melting of the slags. Desired compositions of the slag are given in Table 2. These slags represent an exemplary composition of low basicity refining slag, as used in secondary metallurgical ladle furnace covering slags after Si-/Mn-deoxidation. Similar compositions have been used in prior work, where an improved dissolution of oxidic particles like Al2O3, Al2MgO4, or SiO2 has been shown [22,23].
These premelted slags were powdered to enhance homogeneity. Modification experiments were carried out using an argon-flushed resistance-heated tube furnace (Ruhrstrat HRTK 32 Sond.) with very low O2 partial pressure. ZrO2 crucibles were used in this set of experiments, as other crucible materials, like Al2O3, are prone to deterioration in contact with alkali oxide-containing slags, as shown in previous work by the authors [22,23]. Steel samples of approximately 300 g of a medium-carbon steel, corresponding to material number 1.0623, further called steel 1, were heated to 1600 °C with 15 g (5wt.%) of slag. The system was held at this temperature for 15 min before slow-cooling in the furnace. No stirring or bubbling was possible in this experimental setup. After cooling, steel samples were prepared for automated Scanning Electron Microscopy/ Energy Dispersive Spectroscopy (SEM/EDS) analysis using a JEOL FE-SEM 7200F microscope equipped with a 100 mm2 EDS detector.
3.2 Alkali-carbonate modification experiments
With the second set of experiments, the modification of oxidic NMIs should be achieved by additions of carbonatic compounds to the liquid melt. With this method, the contact between steel and alkali elements should be enhanced, as due to the thermal decomposition of the carbonates, as stated in equation (1), the melt is stirred by the formation of CO2 bubbles. This in addition with a more direct contact of the alkali compound with the melt, should drastically increase the kinetics of reactions between the alkali elements, the melt, and therefore the NMIs. Samples from the same steel grade as in slag modification experiments (steel 1) were heated to 1600 °C using an induction furnace (Indutherm MU 900). This furnace was chosen as it features a large carbon crucible as a heating chamber, which is resistant to attack from alkali compounds in case the inner zirconia crucible breaks in the course of thermal decomposition reactions of the alkali carbonates. As in the previous experiments, zirconia crucibles were used to avoid dissolution of the crucible. Alkali-carbonates had to be prepared before addition to prevent the compound from decomposing on the surface of the steel, but to be successfully deposited below the surface of the steel melt. For this, powdered alkali-carbonates were compressed with pure iron granulate to increase the density and protect the material from fast decomposition. The amount of carbonates was chosen so that after decomposition to the respective oxide, the weight of alkali oxide should amount to 1 wt.% of the steel sample. For the pellet to stick together and to ensure a proper density of the pellet and, therefore, allow for the pellet to sink into the melt, pure iron granules, amounting to approximately 10 times the weight of the carbonates, were used. Ratios of 1:1 as suggested by Chen et al. [18] and Zhao et al. [20] could not be reproduced, as the high relative amount of powdered carbonates prevented the formation of solid pellets. The carbonatic material was surrounded by pure iron granules and pressed using a hydraulic press into a pellet with approximately 100 t of force. This layering of granules and powders is shown schematically in Figure 2.
This procedure leads to a total pellet weight of about 500 g of material, which was added to 250 g of steel. Due to this high percentage of material added during the experiments, the samples were prone to solidification right after the addition of the pellet. After further heating, this was quickly resolved, and thermal decomposition of the carbonates led to intensive mixing of the sample due to CO2 gas formation. This reaction leads additionally to considerable mixing and splashing of the melt. Further, the CO2 reacts with the dissolved carbon in the melt, forming CO gas according to the boudouard reaction as shown in equation (2). This is reducing some amounts of the formed alkali-oxides to its metallic counterpart (Eq. 3), which in turn gasifies and is burned off in a bright flame with a characteristic color for the respective alkali metal at the top of the furnace. Due to this violent reaction of the added alkali-carbonates with the liquid metal, leading to significant material losses, and associated safety concerns, this method was deemed unsuitable for practical application at a larger scale and was not pursued further for inclusion analysis.
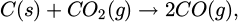 (2)
(2)
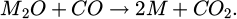 (3)
(3)
 |
Fig. 2 Alkali-carbonate pellet. |
3.3 Combined slag-carbonate modification experiments
The last set of experiments presented is a combination of the experiments described before. Here, in addition to a slag containing alkali oxides, which is used to cover the sample, a pellet, as depicted in Figure 2, containing alkali carbonates is added to the melt at 1600 °C. This procedure ensures protection of the melt from oxidation from the surface, as well as provides a chemical counterpressure to avoid large-scale burn-off of alkali elements from the melt. It was observed during experiments that the otherwise violently strong bubbly behavior that could lead to significant splashing of the melt after the addition of carbonates was not as prominent with the use of a covering slag. Pellets containing alkali-carbonate Na2CO3 or K2CO3, respectively, were produced using the same method as described above. Samples of two different steel grades (steel 1 and another lower carbon steel grade, corresponding to material number 1.7225, further called steel 2) were heated together with powdered slag to 1600 °C. After complete liquefication of steel and slag, the pellet was introduced to the melt. Again, this frequently led to temporary solidification of the melt as the ratio of pellet to melt weight was not negligible. Approximately 5 min after the sample re-liquefied, the melt was stirred using a carbon rod. After an additional 10 min of holding at 1600 °C, the samples were slowly cooled inside the oven until completely solidified. With some samples, increased bubbling during cooling and solidification of the sample could be observed, resulting in a foam- or sponge-like structure. In these cases, samples were molten one additional time to decrease the amount of bubbles in the solidified sample and increase the sample’s quality for the following analysis. After complete cooling of the samples these were again prepared for and subjected to manual and automated SEM/EDS analysis.
4 Results
In the following, the results of the slag treatment as described in chapter 3.1, as well as combined slag-carbonate experiments described in chapter 3.3, are summarized. The inclusion landscape was affected by both methods, but direct modification of inclusions with alkali elements could only be shown using the combined slag-carbonate method.
4.1 Indirect modification of NMIs
The overall landscape of NMIs was changed in all experiments using either only slag or a combination of slag and direct addition of alkali-carbonates. Looking at the relative percentages of oxides, sulfides, and multiphase particles comprising oxidic as well as sulfidic parts, is one of the most effective ways of comparing different inclusion landscapes, especially when normalized to the total area of measured NMI in a reference sample. Particles are classified by their chemical composition, where the content of non-metallic bonding partner (O, S, N) defines the class (oxides, sulfides, nitrides, or combinations of those). Further, particles are additionally typified by looking at the amounts of metallic bonding partners, such as Al, Si, Ca, Mn, among other elements.
Experiments using only slags, carried out using the slow-cooling vertical tube-furnace as described in chapter 3.1, show a significant change in the inclusion landscape of steel 1. After treatment with slag REF 36% of measured particle area was classified as sulfides by automated SEM/EDS analysis. Treatment with slag N5A5 in the vertical tube furnace led to a reduction in the relative area of sulfidic particles to only 2% of the measured area as compared to the steel treated with slag REF. This could be linked to the slow-cooling conditions, as the solubility of sulfur in the slag increases with the addition of Na2O to the slag as shown by thermodynamic calculations. Slow-cooling could possibly leave enough time for S to diffuse from liquid steel into the slag phase, which in turn could lead to lower levels of sulfides forming in the steel due to a lack of available S. Experiments conducted using the faster cooling furnace set-up as described in chapters 3.2 and 3.3 do also show a decrease in the relative area of measured sulfides, but the effect is the same, with and without additions of alkali elements to the system.
In Figure 3 subplot a) shows the relative inclusion landscape after treatment with slag REF, while subplot b) shows the results after treatment with slag N5A5, where a drastic decrease in the relative amount of sulfidic area is shown, while the amount of measured oxidic area increases. However, as shown in previous work, the overall total number of inclusions decreased after treatments with Na-containing slags [22]. Subplots c) to e) show inclusion landscape of combined slag-carbonate experiments carried out in the faster cooling induction furnace, again relative to the NMI-landscape of sample a). Subplot c) shows the relative area of NMI-classes in steel 1 after treatment with slag REF and addition of a pellet solely containing pure iron powder, while subplots d) and e) show the relative inclusion landscape after treatment with slag N5A5 and Na2CO3, or K5A5 and K2CO3, respectively. Here, it becomes apparent that with faster cooling after combined slag-carbonate experiments, the change in the area of OS particles is similar to the difference between samples a) and b), but the relative area of sulfides is not affected by the addition of alkali-bearing components during fast-cooling. The relative area of measured sulfides is 13% for sample c), 12% for sample d), and 14% for sample e), showcasing that only with slow-cooling the desulfurizing effect of alkali-bearing slags can take full effect, while with fast-cooling conditions no such effect could be observed. The decrease in sulfides observed in samples c)-e) is thereby soley contributed to the faster cooling conditions as compared to samples a) and b).Comparing the number of inclusions present in steels 1 and 2 after combined slag-carbonate experiments it was observed that relative to the respective reference experiment the number of inclusions decreased, both with additions of Na-bearing components as well as the addition of K-bearing components. For steel 1 the number of NMIs decreased to 67% when treated with slag N5A5 and Na2CO3 compared to treatment with slag REF, while treatment with slag K5A5 and K2CO3 led to a decrease to 84% of NMIs. In contrast, with steel 2 treatment with K-bearing components proved more effective as the number of NMIs dropped to 56% compared to treatment with slag REF, whereas treatment with slag N5A5 and Na2CO3 only decreased the number of NMIs to 91%.
Regarding the mean and median ECD of observed NMIs no significant change could be found in steel 1 and 2 after combined slag-carbonate treatments in comparison to the samples treated with slag REF and after fast-cooling.
Additionally, a change in the NMI landscape can be observed when comparing the metallic bonding partner of inclusions. This can be visualized by plotting the relative contents of Al, Si, and Mn, which are the most common metallic constituents of observed NMIs in ternary diagrams. In Figure 4, the results of combined slag-carbonate experiments with both steel 1 and steel 2 are shown. In both steels, a distinct change in the chemical composition of NMIs can be observed after combined slag-carbonate experiments. Sub-plots a)- c) show the chemical composition of NMIs detected in steel 1 after treatment with a) reference slag (without contents of Alkali elements), b) N5A5 slag and Na2CO3, and c) K5A5 slag and K2CO3. Sub plots d)- f) show the results of a second steel grade after the respective treatment. In both cases, it can be shown that the combined slag-carbonate treatment leads to less spread of the chemical composition in the ternary system of Al-Mn-Si, and the chemical compositions of NMIs in steels 1 and 2 after alkali-treatment look similar, albeit with a very distinct distribution after treatment with reference slag. A shift towards lower relative amounts of Al is apparent in both steels after treatment with alkali-bearing components as shown in sub-plots b), c), e), and f) as compared to sub-plots a) and d).
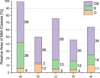 |
Fig. 3 Relative area of NMI-classes in steel 1 after treatment with a) slag REF and slow-cooling, b) slag N5A5 and slow-cooling, c) slag REF, pure iron pellet, and fast-cooling, d) slag N5A5, Na2CO3 pellet, and fast-cooling, e) slag K5A5, K2CO3, and fast-cooling. Data is normalized to total area of measured NMI in sample a). |
 |
Fig. 4 Chemical composition of NMIs in samples of steel 1 after treatment with a) slag REF, b) slag N5A5 and Na2CO3, c) slag K5A5 and K2CO3, Chemical composition of NMIs in samples of steel 2 after treatment with d) slag REF, e) slag N5A5 and Na2CO3, f) slag K5A5 and K2CO3. |
4.2 Direct modification of NMIs
As results for indirect modification of the NMIs in the steel samples with the aid of alkali elements are already shown in the previous chapter, the following results of direct modification of NMIs by Na and K addition are presented. In experiments, using slag as the only alkali-element-bearing component, no directly modified particles (meaning Na- or K-containing) could be found during automated or manual SEM/EDS analysis. Only after combined steel slag experiments was it possible to observe particles containing amounts of Na or K, respectively. A complex oxidic particle containing Al, Si, Mn as well as amounts of Na is exemplarily shown in Figure 5, where throughout the whole particle a homogeneous amount of Na was measured. Most particles containing amounts of Na analyzed manually in the course of this work show similar features, such as a round appearance and a homogeneous chemical composition.
Upon analysis of the chemical compositions of detected NMIs after combined slag-carbonate experiments with Na-bearing components, it was found that Na was mostly prominent in particles also containing Al. This becomes apparent when the relative amounts of metallic components of the particles are plotted in a ternary diagram. In Figure 6, the relative amounts of Al, Si, and Mn measured in particles detected in samples of steel 2 after combined slag-carbonate treatment are shown. Plotted in red on the left-hand side are relative compositions of Na-containing particles, whereas on the right side, the relative chemical composition of particles containing no amounts of Na is depicted in green. Particles near the Mn-corner of the ternary plot tend to show no amounts of Na. This can be explained by the nature of these Mn-rich NMIs, as most of them are found to be purely sulfidic MnS particles. During manual and automated analysis, no Na-modified particles with only S as their non-metallic bonding partner could be observed, as Na was only measured in combination with amounts of O. NMIs with higher amounts of Si or Al tend to also contain Na, which could also be observed by manual analysis and elemental mapping of those particles using EDS. Similar results were found for samples of steel 1.
Figure 7 shows the backscatter electron image of an oxidic particle found in steel 1 after combined slag-carbonat treatment with K-bearing components. Again, amounts of K were found homogeneously throughout the whole particle. Also, other constituents of this particle, Al, Mn, Si, and Ca were homogeneously present in this and other K-modified particles. Like with Na-modified particles, there is a significant trend to favor modification of Al-containing NMIs. Figure 8 shows ternary plots for the relative amount of Al, Si, and Mn in K-modified and non-modified particles. Here, again, particles located near the Mn-rich corner are less likely to be modified as these particles are mainly sulfidic, whereas particles with higher amounts of Al and Si often show amounts of K present.
Another example of the affinity of K for Al is presented in Figure 9. Here a complex particle containing amounts of Al, Mn, Si, and K is depicted. In this case the chemical composition of the particle is not homogeneous. The particle features multiple distinct phases. The darkest areas contain amounts of Al, Si, and K, whereas the lighter outer area shows amounts of Si and Mn, but contains neither Al nor K. The observed selective modification of Al-rich phases by Na and K may be related to the thermodynamic calculations presented in Section 2, which indicate that the presence of Na2O and K2O lowers the liquidus temperature of Al2O3. However, the underlying mechanisms governing alkali modification of NMIs are not yet fully understood. Further research will therefore focus on clarifying the mechanistic pathways responsible for these modification processes.
 |
Fig. 5 Backscatter Electron Image of a complex oxidic particle with elemental mappings of Al, Si, Mn, and Na as present in steel 1 after respective combined slag-carbonate treatment. |
 |
Fig. 6 Comparison of chemical composition of a) Na-modified and b) non-modified NMIs in samples of steel 2 after combined N5A5 slag Na-carbonate treatment. |
 |
Fig. 7 Backscatter Electron Image of a complex oxidic particle with elemental mappings of Al, Si, Mn, and K as present in steel 1 after respective combined slag-carbonate treatment. |
 |
Fig. 8 Comparison of chemical composition of a) K-modified and b) non-modified NMIs in samples of steel 2 after combined K5A5 slag K-carbonate treatment. |
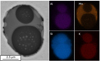 |
Fig. 9 Backscatter Electron Image of a complex multiphase oxidic particle with elemental mappings of Al, Si, Mn, and K as present in steel 1 after respective combined slag-carbonate treatment. |
5 Discussion and conclusion
The present study explored different strategies for introducing alkali elements into liquid steel during secondary refining, with the aim of evaluating their potential for NMI control. The experimental matrix, comprising alkali-oxide bearing slags, alkali-carbonate additions, and combined slag—carbonate treatments, provides a systematic view of both indirect and direct inclusion modification. The analysis by manual and automated SEM/EDS enabled a quantitative and qualitative assessment of the inclusion landscape in two steel grades after respective refining treatments. One key finding was that carbonate addition without the protection of a covering slag layer is not suitable for refining practice on a laboratory scale. The uncovered treatment led to intense splashing of the melt and a pronounced burn-off of alkali elements, which not only reduces process efficiency but also poses safety and yield concerns. This observation underscores the necessity of a slag layer to stabilize the reaction kinetics and minimize uncontrolled volatilization losses of alkali components.
With respect to inclusion control, the results clearly indicate that indirect modification of inclusions is achievable through both alkali-bearing slags and combined slag-carbonate treatments. Particularly, a consistent trend towards reduced relative amounts of sulfide inclusions was observed. The observed decrease suggests that the alkali additions influence the solubility of sulfur in steel, slag, and sulfide phases, which has also been shown by thermodynamic calculations. This results in a strong dependence of observed sulfide amounts on the cooling conditions during treatment of the steel with alkali element-containing components.
The experiments also highlight that direct modification of inclusions by Na and K requires the presence of both slag and carbonate sources. Slag treatment alone was not sufficient to achieve this effect. When both sources were present, however, direct alkali modification of oxidic NMIs was confirmed. Na and K showed a strong preference for modifying Al-rich inclusions. Particularly relevant is the finding that Na and K modifications occur not only in monophase alumina particles but also selectively within alumina phases of multiphase inclusions. As thermodynamic calculations could show, this should in theory aid in decreasing the liquidus temperature of these directly modified Al-containing NMIs. Modification with Na or K of sulfidic phases of NMIs could not be observed.
If Na and K additions can reliably modify the chemical composition of Al-rich NMIs, significant improvements in steel cleanliness and performance could be realized.
Overall, the study demonstrates both opportunities and limitations of alkali-based inclusion engineering. Indirect effects via slag chemistry appear promising for reducing sulfide populations, while direct modifications of alumina inclusions require combined addition strategies. These findings provide a valuable foundation for tailoring refining practices to exploit alkali effects on secondary refining practices and steel cleanness. Future work will focus on quantifying the behavior of alkali-modified NMIs, particularly their deformation characteristics during forming, as well as optimizing slag–carbonate ratios to balance efficiency, stability, and safety.
Funding
The financial support by the Austrian Federal Ministry of Economy, Energy and Tourism, the National Foundation for Research, Technology and Development, the Christian Doppler Research Association and voestalpine Stahl Donawitz GmbH is gratefully acknowledged.
Conflicts of interest
The authors have nothing to disclose.
Data availability statement
Data associated with this article cannot be disclosed due to legal reasons.
Author contribution statement
Conceptualization, N. Preisser, G. Klösch, and S. K. Michelic.; Methodology, N. Preisser; Software, N. Preisser; Validation, N. Preisser, B. Suppan, G. Klösch, and S. K. Michelic.; Formal Analysis, N. Preisser and B. Suppan; Investigation, N. Preisser and B. Suppan; Resources, G. Klösch and S. K. Michelic; Data Curation, N. Preisser and B. Suppan; Writing – Original Draft Preparation, N. Preisser; Writing – Review & Editing, N. Preisser, G. Klösch and S. K. Michelic; Visualization, N. Preisser; Supervision, S. K. Michelic; Project Administration, G. Klösch and S.K. Michelic; Funding Acquisition, G. Klösch and S. K. Michelic.
References
- L. Zhang, B.G. Thomas, State of the art in evaluation and control of steel cleanliness, ISIJ Int. 43.3, 271 (2003) [Google Scholar]
- C. Chen, Z. Jiang, Y. Li et al., State of the art in the control of inclusions in tire cord steels and saw wire steels – a review, Steel Res. Int. 90.8, 1800547 (2019) [Google Scholar]
- C. Chen, Z. Jiang, Y. Li et al., Effect of Rb2O on inclusion removal in C96V saw wire steels using low-basicity LF refining slag, ISIJ Int. 58.11, 2032 (2018) [Google Scholar]
- C. Chen, M. Sun, X. Chen et al., State of the art in control of inclusions and microalloying elements in tire cord steel and saw wire steel, Steel Res. Int. 93.4, 2100507 (2022) [Google Scholar]
- Q. Shu, C. You, T. Alatarvas et al., Experimental and modeling study of deformability of glassy CaO-(MnO)-Al2O3-SiO2 inclusions, Metals 12.3, 522 (2022) [Google Scholar]
- L. Zhang, C. Guo, W. Yang et al., Deformability of oxide inclusions in tire cord steels, MMTB 49.2, 803 (2018) [Google Scholar]
- C. Chen, Z. Jiang, Y. Li et al., State of the art in the control of inclusions in spring steel for automobile - a review, ISIJ Int. 60.4, 617 (2020) [Google Scholar]
- U. Karr, Y. Sandaiji, R. Tanegashima et al., Inclusion initiated fracture in spring steel under axial and torsion very high cycle fatigue loading at different load ratios, Int. J. Fatigue 134, 105525 (2020) [CrossRef] [Google Scholar]
- Y. Furuya, T. Abe, S. Matsuoka, 1010-cycle fatigue properties of 1800 MPa-class JIS-SUP7 spring steel, Fatigue Fract. Eng. Mater. Struct. 26.7, 641 (2003) [Google Scholar]
- A. Srivastava, A. Kamaraj, D. Mandal et al., Role of synthetic slag treatment on the morphology of non-metallic inclusions and subsequent cold drawability of the high carbon wire rod steel, Met. Mater. Int. 28.7, 1763 (2022) [Google Scholar]
- K. Choi, Y. Kang, I. Sohn, Effect of Rb2O and Cs2O on inclusion removal in 321 stainless steels using novel basic tundish fluxes, MMTB 47.3, 1520 (2016) [Google Scholar]
- Y. Li, C. Chen, Z. Jiang et al., Application of alkali oxides in LF refining slag for enhancing inclusion removal in C96V saw wire steel, ISIJ Int. 58.7, 1232 (2018) [Google Scholar]
- J.Y. Yu, Y. Kang, I. Sohn, Novel application of alkali oxides in basic tundish fluxes for enhancing inclusion removal in 321 stainless steels, MMTB 45.1, 113 (2014) [Google Scholar]
- J. Zhouhua, C. Chen, Y. Li et al., Effect of K2O on inclusion removal in C96V saw wire steels using LF refining slags, The challenge of industry 4.0., in: Proceedings of International Congress on Science and Technology of Steelmaking, AIM Associazione italiana di metallurgia, Milano, Italy, 2018. [Google Scholar]
- L. Chen, W. Chen, W. Yan et al., Refractory/steel/inclusion interactions in Al-deoxidised valve spring steel treated with Na 2 CO 3, Can. Metall. Q. 36, 58.3 (2019) [Google Scholar]
- Z. Zhao, Y. Wang, D. Hou et al., Investigation on the deformation mechanism of alkali metal on inclusions in cord steel containing different aluminium content, Ironmak. Steelmak. (2024) [Google Scholar]
- C. Chen, Z. Jiang, Y. Li et al., Effect of Na2O and Rb2O on inclusion removal in C96V saw wire steels using low-basicity LF (Ladle Furnace) refining slags, Metals 8.9, (2018) [Google Scholar]
- L. Chen, W. Chen, Y. hu et al., Effect of Na2CO3 addition on inclusions in high-carbon steel for saw wire, Trans. Indian Inst. Met. 71.2, 383 (2018) [Google Scholar]
- L. Chen, Y. Wan, J. Li et al., A new method for plasticization of inclusions in saw-wire steel by NaF addition, Metals 10.6, 704 (2020) [Google Scholar]
- J. Zhao, Y. Wang, M. Wang et al., Effects of Na2O, K2O and B2O3 on deformability of SiO2-MnO-Al2O3 inclusion in high-carbon steel, Metals 13.8, 1341 (2023) [Google Scholar]
- C. W. Bale, E. Belisle, P. Chartrand et al., FactSage thermochemical software and databases – 2010–2016, Calphad 54, 35 (2016) [CrossRef] [Google Scholar]
- N. Preisser, J. Cejka, S. Ramesh Babu et al., Dissolution of Al2O3, MgO•Al2O3 and SiO2 in alkali oxide containing secondary metallurgical slags, in: Proceedings of the Liquid Metal Processing & Casting Conference, The Minerals, Metals & Materials Society, Philadelphia, USA, 2022, pp. 87–96 [Google Scholar]
- N. Preisser, J. Cejka, K. Thiele et al., Investigation of inclusion evolution in a medium carbon steel through contact with secondary metallurgical slags containing alkali oxides, in: Proceedings of METEC & 6th ESTAD 2023, Steel Institute VDEh. Dusseldorf, Germany, 2023 [Google Scholar]
Cite this article as: Nikolaus Preisser, Benjamin Suppan, Gerald Klösch, Susanne K. Michelic, Comparison of methods for modification of non-metallic inclusions using alkali elements on a laboratory scale, Metall. Res. Technol. 123, 209 (2026), https://doi.org/10.1051/metal/2025138
All Tables
Desired slag compositions and composition as measured by XRF (in parentheses), where M2O stands for Na2O or K2O, respectively.
All Figures
 |
Fig. 1 Solubility of Sulfur in steel-, slag, and sulfide phase in contact with a) Na2O-containing slag, b) K2O-containing slag. |
| In the text | |
 |
Fig. 2 Alkali-carbonate pellet. |
| In the text | |
 |
Fig. 3 Relative area of NMI-classes in steel 1 after treatment with a) slag REF and slow-cooling, b) slag N5A5 and slow-cooling, c) slag REF, pure iron pellet, and fast-cooling, d) slag N5A5, Na2CO3 pellet, and fast-cooling, e) slag K5A5, K2CO3, and fast-cooling. Data is normalized to total area of measured NMI in sample a). |
| In the text | |
 |
Fig. 4 Chemical composition of NMIs in samples of steel 1 after treatment with a) slag REF, b) slag N5A5 and Na2CO3, c) slag K5A5 and K2CO3, Chemical composition of NMIs in samples of steel 2 after treatment with d) slag REF, e) slag N5A5 and Na2CO3, f) slag K5A5 and K2CO3. |
| In the text | |
 |
Fig. 5 Backscatter Electron Image of a complex oxidic particle with elemental mappings of Al, Si, Mn, and Na as present in steel 1 after respective combined slag-carbonate treatment. |
| In the text | |
 |
Fig. 6 Comparison of chemical composition of a) Na-modified and b) non-modified NMIs in samples of steel 2 after combined N5A5 slag Na-carbonate treatment. |
| In the text | |
 |
Fig. 7 Backscatter Electron Image of a complex oxidic particle with elemental mappings of Al, Si, Mn, and K as present in steel 1 after respective combined slag-carbonate treatment. |
| In the text | |
 |
Fig. 8 Comparison of chemical composition of a) K-modified and b) non-modified NMIs in samples of steel 2 after combined K5A5 slag K-carbonate treatment. |
| In the text | |
 |
Fig. 9 Backscatter Electron Image of a complex multiphase oxidic particle with elemental mappings of Al, Si, Mn, and K as present in steel 1 after respective combined slag-carbonate treatment. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


