| Issue |
Metall. Res. Technol.
Volume 123, Number 2, 2026
Special Issue on ‘Innovations in Iron and Steelmaking’, edited by Carlo Mapelli and Davide Mombelli
|
|
|---|---|---|
| Article Number | 211 | |
| Number of page(s) | 9 | |
| DOI | https://doi.org/10.1051/metal/2025152 | |
| Published online | 11 February 2026 | |
Original Article
Concepts for the development of carbon-free mold powders for the continuous casting of steels
Technical University of Leoben – Chair of Ceramics, Peter Tunner Straße 5, 8700 Leoben, Austria
* e-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
20
August
2025
Accepted:
25
November
2025
Abstract
Mold powders for the continuous casting of ultra-low-carbon steels already contain low amounts of carbon. Furthermore, various approaches favor carbon burn-off at high temperatures to prevent the formation of a carbon-enriched layer on top of the liquid slag pool in the mold. Nevertheless, re-carburization of the steel still occurs. Therefore, the development of different mold powder compositions to reduce the free carbon content without negatively affecting melting behavior is required. An initial approach involved replacing carbon with SiC, which showed promising results. Subsequently, further laboratory investigations were conducted on different mold powder compositions with reduced SiC contents. However, to further decrease the total carbon content, a new concept is required: a mold powder consisting of basic and acidic granules, which are mixed after granulation. In the next step, melt-controlling additives are completely removed from the samples. For these mold powders, the influence of varying raw material components on melting behavior was additionally investigated. To verify this concept, the standard and newly developed mold powders were introduced into a furnace preheated to temperatures between 900−1200°C to simulate high heating rates. After a dwell time of 10 min followed by quenching to room temperature, the samples were investigated mineralogically. The results reveal that the diffusion path between raw material particles is increased due to the separation of the raw materials into two different granule types (basic and acidic). Furthermore, the formation of an intermediate liquid phase is shifted to higher temperatures. This shift results in a delay in the formation of new (intermediate) phases, such as the equilibrium liquid phase, fluorides, or cuspidine. The developed concept enables the complete removal of melt-controlling additives from the mold powder composition. As a side effect, the CO2 emissions from the mold powder is reduced.
Key words: mold powder / carbon-free / melting behavior / two types of granules
© EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Mold powder is added to the liquid steel meniscus in the mold during continuous casting. To ensure stable casting conditions, it fulfills several tasks, e.g., preventing steel from oxidation, lubrication of the strand, and controlling horizontal and vertical heat transfer. When added to the mold on top of the liquid metal, three layers are formed due to the temperature gradient between the steel to the atmosphere. In contact with the air, the mold powder is present in the state as-delivered. With increasing temperature, a sintered layer is formed, and in contact with the steel, a liquid slag exists [1]. The melting behavior of the mold powder is controlled by different carbon carriers added to the mixture. At low temperatures the solid – solid reactions between raw material particles are prevented. This is mainly controlled by carbon black with a low particle size. Carbon black effectively encloses the particles and separates them. In contrast, at high temperatures it is already oxidized, whereas graphite prevents the formation of a continuous liquid phase by separating liquid droplets. This phenomenon is known as the skeleton effect [2–5]. Generally, it is intended that the total amount of carbon is burned off until the slag comes into contact with the liquid steel. However, this depends on the oxygen supply and may not be completed. In this case, a carbon-enriched slag layer is formed on top of the liquid slag pool. Its carbon content is considerably increased, e.g., from 3.5 wt% in the as-delivered state to 21 wt% [5,6]. Similar results were also obtained in laboratory investigations [7,8]. If the liquid steel comes into contact with the carbon-enriched layer, re-carburization takes place. Subsequently, steel properties are altered and product quality is reduced. This is especially the case for ultra-low-carbon steels [8]. Thus, to increase the distance between the liquid steel and the carbon-enriched layer, higher slag film thicknesses are desired. Another possibility to prevent this reaction is the complete oxidation of carbon until a homogeneous liquid phase is present. This can be achieved by adding oxidizing agents such as MnO2, by using quick-burning carbons (activated carbons), or by the application of Fe2O3, which acts as a catalyst for carbon oxidation and is also reduced by carbon at high temperatures and low oxygen partial pressures [9–12].
Further developments were achieved by replacing free carbon as the melt-controlling additive in the mold powder [13–16]. For this purpose, ceramic materials showing poor wettability with slags were selected. In a first step, different nitrides were tested, and BN showed the most promising results. This was explained by its similar structure to carbon and its high contact angle with slags. Furthermore, it showed a low gas evolution during heating in laboratory tests and controlled the melting behavior of the mold powder similarly to carbon. The minimum amount required in the product to control melting is defined by the particle size of all raw material components. When comparing the fusion rate of a BN containing mold powder with a standard carbon-containing mold powder, similar results were obtained for samples containing 2−10 wt% BN. As BN forms B2O3 after oxidation, which acts as a fluxing agent and favors sintering, antioxidants (e.g., Al or CaSi) were added to shift liquid phase formation to higher temperatures. Boron oxide also influences slag viscosity. Thus, proper viscosity values were only achieved for samples with a CaO/SiO2 ratio of at least 0.9. Based on these results, a mold powder was selected for industrial trials with defined steel grades. The slabs showed the incorporation of N into the liquid steel decreasing its quality, because nitrogen pick-up is as detrimental as carbon pick-up. As a result, the amount of BN has to be reduced, but this is only possible if the total amount of carbon is not removed [8,13,14]. Contrary, Si3N4 forms SiO2 after oxidation, a component already present after liquefaction of standard mold powders. Furthermore, it shows a low sintering tendency. An average particle size of 5 μm and a specific surface area of 2.5−3.5 m2 g−1 was used for the investigation of mold slag compositions in the laboratory. Nevertheless, it was suitable for controlling powder melting, nitriding of the steel was observed in slabs when used in the continuous casting [15–18].
Attempts to produce color-coded mold powders for different steel grades required the development of mold powders without free carbon. When investigated with the heating microscope similar temperatures for the softening, melting, and flowing points were achieved as for the carbon-containing reference material, but the melting rate increased. The authors claim that the continuous casting process is not negatively affected as long as a layer of original powder is still present in contact with the atmosphere [19].
In addition, studies using SiC as a melt-controlling component instead of carbon were carried out. Comparing a sample containing 5−6 wt% SiC with a standard mold powder containing 2−2.5 wt% free carbon, similar melting rates were observed. Nevertheless, the three layers differed: Whereas only a very thin layer of loose powder was present, the sintered layer increased due to SiC oxidation, forming SiO2 and thus decreasing the basicity of the flux and promoting liquid phase formation. For this reason, carbon was not completely substituted. Industrial trials showed that re-carburization still takes place if the carbon content exceeds 1 wt% [17,20–23].
Although these newly developed mold powders showed promising results in industrial trials, they have not prevailed yet. Possible reasons include the increased price of these mold powders, which may not justify the benefits, or a lack of confidence in their potential. Nevertheless, steel plants still face the problem of re-carburization [24–27], but no further research on mold powder design has been reported in recent years. Thus, it is necessary to drive development forward again. As a first step, to investigate the influence on melting behavior beyond a purely phenomenological approach, a mold powder used for ultra-low-carbon steel was selected [28]. Based on its chemical composition, different mixtures without free carbon were calculated without changing the overall chemical composition of the slag formed after total liquefaction. SiC and/or Si3N4, with or without antioxidants, were used to control the fusion rate. After temperature treatment in a preheated muffle furnace, ensuring high heating rates, the samples were quenched to room temperature after the defined dwell time and investigated mineralogically. It was evident that both additives shifted the formation of new solid and equilibrium liquid phases to higher temperatures, but SiC proved to be the most efficient. This is explained by the stability of SiC. Below 1100°C, it is still present in the solid state, and SiO2 is not available for new phase formation. Thus, reactions are delayed. The amount of SiC could be reduced to 2.8 wt% [28,29], which is even below the amount suggested in the literature [23], without negatively affecting the fusion rate. Furthermore, antioxidants [30–32] were added to improve the effect of SiC. Although their positive effect was confirmed by thermodynamic calculations, the samples showed liquid phase formation at lower temperatures. Additionally, due to the rapid oxidation of the antioxidants, the amount of liquid phase was increased. Consequently, for mold powder compositions, only the addition of SiC was suggested. Based on these results, it was concluded that reactions between raw material particles must be delayed to reduce the melting rate [28,29]. Nevertheless, further decrease of melt-controlling additives is only possible if the physical distance between phases that form new phases by solid – solid diffusion at lower temperatures is increased. This approach may be realized by dividing the raw material particles into two groups: one representing the basic components and the other the acidic components. Basic and acidic granules are merged into one product according to the chemical composition of the desired mold powder composition. The resulting physical distance between phases of two contacting basic and acidic granules of spherical shape and equal size, with an arrangement in which one granule type is surrounded by granules of the other type only, depends on the location within the respective granules. It starts at 0 mm at the point of direct contact between basic and acidic granules and increases up to the diameter of a single granule (0.5−1 mm). Therefore, the aim of this study is to verify retardation of liquefaction using this concept of two different granule types. This is achieved by investigating the melting behavior of various mold powder samples containing basic and acidic granules made from different raw material components and comparing them with the standard mold powder. Additionally, the influence of different raw materials on the temperature-dependent stability of the respective granules is examined.
2 Material and methods
A mold powder (MP 0) used for casting low-carbon steels and containing 3.78 wt% of total carbon was selected. Based on the chemical composition of the mold powder, the theoretical slag composition present after liquefaction (Tab. 1) was calculated, raw materials were selected, and the mixture was determined. All samples exhibit identical chemical compositions to the first decimal place, including trace elements. This corresponds to a deviation of less than 0.1%. Where reasonable, similar raw material components similar to those present in the standard mold powder were used. The main raw materials of MP 0 are wollastonite (CaSiO3), albite (NaAlSi3O8), and fluorite (CaF2). Quartz (SiO2), magnesite (MgCO3), calcite (CaCO3), hematite (Fe2O3), diopside (CaMgSi2O8) as well as synthetic raw materials such as phosphor slag and blast furnace slag are used in lower amounts. Minor phases are carbon black (C), cryolite (Na2AlF6), dolomite (CaMg(CO3)2), and Ca2SiO4. Hematite serves as a Fe2O3 source in MP 0 as well as in the mixtures investigated in this study to achieve the desired properties during casting, e.g., by acting as a catalyst for carbon oxidation [11].
In the next step, depending on its chemical composition, each raw material was assigned either to the basic or the acidic group. Wollastonite was assigned to the acidic components in this study due to its CaO/SiO2 ratio of 0.93. Furthermore, raw materials consisting mainly of amphoteric oxides and fluorite were also assigned to the acidic group. The raw material compositions of the investigated samples are summarized in Table 2. First, to evaluate how the separation of the raw material particles into two groups affects the melting behavior of the mold powder, a composition containing SiC was chosen (MP 1). This sample was already investigated in previous studies in a powdery state, without separating the raw material particles into basic and acidic groups [28,29]. In the next step, the amount of SiC in the acidic granules was reduced. Previous studies have revealed that the melting behavior of mold powders is also affected by their raw material composition [4,33,34]. Thus, two mixtures differing in raw material composition were calculated, and their liquefaction behavior was investigated. Furthermore, based on the promising results of MP 2 and MP 3 in Section 3.2, the melt-controlling additive SiC was completely removed. The purpose was to verify the assumption that melt-controlling additives can be completely extracted from the mold powder composition if the contact of possible reactants is considerably reduced through the use of two different granule types in a mold powder (MP 4 and MP 5).
The basic and acidic mixtures were prepared, homogenized in a disc mill, and with the help of a bottle roller (filling level > 50%) granules were produced. As a binder, starch dissolved in water (2% solution) was used and sprayed onto the mixture particles during granulation. After granulation, the basic and acidic granules were dried and sieved. Granules with particle sizes between 0.5 and 1 mm were used for the investigations to ensure similar sizes in both granule types. The acidic and basic granules were weighed according to the desired chemical composition of the mold powder (Tab. 2). Mechanical mixing of the granules was performed in an airtight polyethylene bag to achieve a uniform distribution of components without destroying the granules. Then, each sample was filled into a separate steel crucible 30 × 30 × 40 mm3, closed by a steel lid and inserted into the preheated furnace. Temperatures of 900°C, 1000°C, 1100°C and 1200°C, respectively, were selected to investigate the sintering tendency and the formation of new solid and liquid phases in dependence on temperature and hence, on the position within the mold powder layer in the mold. 1200°C was selected as the maximum temperature due to the presence of a continuous liquid phase. A further increase in temperature would not reveal any further information. The dwell time was 10 min to ensure complete heating of the sample without reaching equilibrium, before the specimens were taken out from the furnace and naturally cooled to room temperature (approximately 23°C). Afterwards, polished sections were prepared and investigated mineralogically using reflected light microscopy scanning electron microscopy coupled with energy-dispersive X-ray microanalysis. Furthermore, the samples were ground for X-ray analysis.
Mold slag composition after total liquefaction of MP 0 in wt% (The fluorine content is expressed once in elemental form and once in the form of CaF2).
Sample raw material composition in wt%.
3 Results
3.1 Thermodynamic calculation
In a first step, for each granule composition, the solidus and liquidus temperatures, as well as the amount of liquid phase at 1000°C were calculated thermodynamically. For this purpose, the software FactSage 8.3 was applied. If SiC was used as a raw material, it was also considered in the calculations. The respective results are summarized in Table 3 and are compared with those of the standard mold powder composition (MP 0). Under equilibrium conditions, all samples show higher liquidus temperatures for the basic granules than for MP 0. Whereas the liquidus temperatures of the acidic granules for MP 1 – MP 3 are also higher, they are lower in the cases of MP 4 and MP 5 containing no SiC. Contrary, the solidus temperatures of basic granules are lower, but higher for the acidic ones when compared with MP 0. These results indicate a decelerated formation of liquid phase during heating. Considering the total amount of liquid phase at 1000°C under equilibrium conditions, only MP 2 and especially MP 3 show lower values than the standard carbon-containing mold powder. Nevertheless, this may not be the case in the mold due to the high heating rate and associated intermediate liquid phase formation in disequilibrium. Thus, annealed specimens are investigated mineralogically in Section 3.2.
If wollastonite, which also contains a basic oxide (CaO), is used as a raw material component in the acidic granules, the liquidus temperature decreases and the amount of liquid phase at 1000°C increases. Nevertheless, wollastonite is a common raw material component in mold powders. Thus, it was intended to use it in the mixture composition for further investigation.
In contrast to these calculations, the equilibrium state is not achieved during melting in the continuous casting process, and reaction kinetics play an essential role. Due to the increased distance between acidic and basic components of the mold powder, it is assumed that the sintering tendencies of the samples are further reduced, and the formation of new phases and the equilibrium liquid phase is shifted to higher temperatures.
Thermodynamically calculated values representing the equilibrium state [°C].
3.2 Mineralogical investigations
To verify the assumption that an increase in physical distance delays the melting rate, initial investigations were carried out on a mold powder composition previously investigated as a single mixture (MP 1). The basic granules contain calcite (CaCO3), magnesite (MgCO3), and sodium carbonate (Na2CO3), the acidic ones quartz (SiO2), albite Na(AlSi3O8), fly ash, wollastonite (CaSiO3), SiC, hematite (Fe2O3) and fluorite (CaF2). The mineralogical investigations reveal that the fine basic particles start sintering already at 900°C and decarburization of the larger carbonate particles is observed at 1000°C. Decarburization of calcite and magnesite takes place between 900−1000°C. Due to the selected dwell time, fine carbonaceous particles are completely decarburized, but the larger ones were exposed to this temperature for a too short period. Thus, decarburization was only detected at the porous appearance of their surfaces, but in the dense center, carbon could still be detected. This result was also confirmed by X-ray diffraction analysis. In contrast, the acidic raw material components are mainly present in their original state. Only the dissociation of fluorite (CaF2) via solid – solid diffusion is detected. Fluorine diffuses into the basic granules and forms magnesium fluoride and a fluorine-containing liquid phase (Fig. 1). Increasing the temperature to 1100°C causes the sintering of basic and acidic granules and the formation of a joined, continuous liquid with large pores. Mainly cuspidine (Ca4(Si2O7)(F, OH)2), SiC and residuals of intermediate fluorine containing phases are present in the acidic granule. At 1200°C due to fast liquefaction of the sample at the surface preventing oxygen supply for SiC oxidation, SiC residuals are still observed at this temperature. Furthermore, large wollastonite particles have not yet been dissolved into the liquid phase and are partly still detected in the sample. In contrast, for the carbon containing standard mold powder an independent liquefaction of each granule is observed which has already been discussed in [28,35].
Comparing these results with those of the standard carbon-containing mold powder, a reduced sintering tendency is observed. Thus, in the next step, the amount of SiC used to control the melting rate was reduced to 2.6 wt%. Furthermore, the effect of different raw material components was investigated for MP 2 and MP 3. Compared to MP 1, the reduction in SiC content results in the formation of new phases at the granule boundaries already at 900°C. Na2O diffuses into the acidic granules, Fluorine into basic ones, forming different fluorides and, marginally cuspidine at the contact zone between the two granule types. Simultaneously, the onset of calcite dissociation is observed. Further rise of temperature to 1000°C results in an increasing amount of liquid phase, especially in the acidic granules; however, large solid raw material particles still exist (Fig. 2). Cuspidine is present at the surface of basic granules and within the acidic granules. The appearances of samples MP 2 and MP 3 after heat treatment at 1100°C and 1200°C are comparable to those of MP 1, having a higher SiC content of 5.99 wt%. In all samples, a continuous liquid phase with large cuspidine crystals is present, containing areas of conglomerated, residual SiC particles.
For a sample consisting only of a single granule type, further reduction of the SiC content is not reasonable because it is the only measure to inhibit reactions between raw material components. In contrast, for mold powders consisting of two different granule types, also the increased distance for diffusion affects their interaction. Based on these considerations, further decrease in SiC content or even the complete removal of any melt-controlling additives, is feasible. To verify this assumption, samples containing no raw material that controls the melting behavior of the mold powder were produced. Thus, the composition of the acidic granules and, subsequently, the basic granules was adapted (MP 4 and MP 5): The acidic ones contain quartz, fly ash, fluorite, wollastonite, corundum, and iron oxide but differ in the addition of blast furnace slag. The raw materials of the basic granules were kept the same, but their amounts was adjusted based on the overall chemical composition of the standard slag after liquefaction. In particular, at 900°C an obvious reaction boundary from the granule surface towards the center is observed. This effect is even more pronounced for the basic granules. An intermediate liquid phase is already present at this temperature in the basic granules; where due to the fluorine diffusion, new fluorine-containing solid phases (e.g., calcium magnesium fluorides or magnesium fluorides of different compositions) and liquid phases are formed. Nevertheless, the amount of liquid phase is still low. In the case of MP 5, a porous rim at the boundary of the basic granules is observed due to the reaction of fine particles; however large particles do not take part in any reaction yet. Only, quartz particles closer to the granule surface reveal a marginal zone showing an increase in basic oxides due to diffusion. As observed in the previously investigated samples, cuspidine starts to form at the contact area between the granules. The samples annealed at 1000°C reveal an increase in Na2O in acidic components and decarburization of carbonates. The latter results in a considerable mass loss of the basic granules and their porous appearance. New phase formation, e.g. of cuspidine or an intermediate liquid phase, is also detected only at the granule surfaces. In most cases, the raw materials in the center are still not affected by any reaction (Fig. 3). If the temperature rises to 1100°C, the amount of liquid phase increases, but the granule shapes are still preserved. Thus, a continuous liquid is not present even though no melt-controlling component was added to this mixture. Whereas for MP 4, all granules seem to have more or less the same mineralogical composition (Fig. 4a), in case of MP 5 (Fig. 4b), partly residues of basic granules may still be detected. To explain the differing behavior, FactSage calculations for the granules were carried out. The results summarized in Table 3 show that the amount of liquid phase in MP 4, especially for the basic granules, is considerably higher than for MP 5. Partly replacement of wollastonite in the acidic granules by blast furnace slag and the by this caused necessary increase of calcite in the basic granules of MP 5, seem responsible for the retarded liquefaction of MP 5 compared with MP4. One may consider this as an increase in the basicity difference between the two granules types. The presence of a liquid phase favors the interaction between raw material particles and the formation of new intermediate phases and additional liquid phase. For the acidic granules of both samples, high amounts of quartz are used. These particles are still present in notable numbers at 1100°C. This indicates their slow reaction tendency and additionally contributes to the fact that MP 4 still shows granular shapes, and the existence of basic granules of MP 5 even at this temperature. Besides quartz, both samples also reveal the presence of wollastonite particles in contact with the liquid phase and cuspidine.
 |
Fig. 1 Backscattered electron image of (a) a basic and (b) an acidic granule of sample 1 after annealing at 1000°C: 1 fluorite, 2 wollastonite, 3 SiC, 4 cuspidine, 5 residuum of magnesite, 6 magnesium fluoride, 7 fluorine-containing liquid phase, 8 albite. |
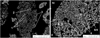 |
Fig. 2 Backscattered electron image of (a) a basic and (b) an acidic granule of sample MP 3 after annealing at 1000°C: 1 magnesium fluoride, 2 calcium magnesium fluoride, 3 cuspidine, 4 liquid phase, 5 fluorite, 6 wollastonite, 7 SiC, 8 albite. |
 |
Fig. 3 Backscattered electron image of (a) MP 4 and (b) MP 5 after annealing at 1000°C: 1 cuspidine, 2 wollastonite, 3 quartz, 4 hematite, 5 fluorite, 6 diopside, 7 Ca2SiO4, 8 calcite, 9 magnesite, 10 calcium magnesium fluoride, 11 magnesium fluoride, 12 liquid phase. |
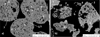 |
Fig. 4 Backscattered electron image of sample MP 4 after annealing at 1100°C: 1 cuspidine, 2 wollastonite, 3 quartz, 4 feldspar, 5 fluorite, 6 diopside, 7 liquid phase, 8 calcium magnesium fluoride, 12 liquid phase. |
4 Discussion
In contrast to the thermodynamic calculations, in the continuous casting process and during the temperature treatment of the samples in laboratory, no equilibrium state is achieved during ongoing liquefaction. Thus, the results of the thermodynamic calculations can only be used to estimate the melting behavior of these samples. Furthermore, acidic and basic components were considered independently of each other for calculating the phase composition as a function of temperature.
The results of the samples containing 5.99 wt% SiC as melt-controlling additives show a considerable reduced melting tendency compared with the standard mold powder. Thus, the reduction of its content was reasonable, which was confirmed by the experimental results. The appearance of the investigated samples with reduced SiC content in dependence on the temperature was similar to that of the standard mold powder. The carbon content given in g/100 g mold powder could be reduced from 1.84 for the standard mold powder (MP 0) to 1.79 for MP 1 and 0.83 for MP 2, respectively. To further reduce the overall carbon content in mold powders and to remove the melt-controlling additives, the reaction kinetics must be influenced using another approach: a mold powder composition containing two different granule types. The investigations revealed that separating basic and acidic raw material components increases diffusion paths. Furthermore, the presence of an intermediate liquid phase is shifted to higher temperatures. These intermediate liquids are formed between components that are adjacent but are not members of a stable paragenesis in chemical equilibrium, e.g., silica (SiO2)and lime (CaO). By separating these components into different granules, such intermediate liquid formation is mitigated. Both effects diminish the reaction rate between acidic and basic components, to form new (intermediate) phases, e.g., different fluorides, cuspidine or liquid phase. It has been observed that reducing the amount of liquid phase in both granule types is associated with retarded liquefaction. It is plausible that a lower melt amount delays chemical equilibration by liquid diffusion. The sum of the separately calculated liquid phase amounts of both granule types will not represent the melt content formed when both granule types interact.
Considering the stability of the basic granules, it is observed that the selection and combination of raw material components further influence the melting behavior of mold powders. This effect is even more evident for mold powders made from two types of granules without melt-controlling additives than for mixtures containing these additives. An increased number of different basic raw material components further supports the basic granule stability with increasing temperature. A high CaO/SiO2 ratio of the mold powder is advantageous because the amount of the basic components is increased. Calcite, showing lower reactivity than, e.g., soda, supports the stability of basic granules when added in larger quantities. The temperature of liquid phase formation is shifted to higher temperatures, and the interactions between basic and acidic raw material components are reduced, which leads to an increase of the stability of the basic granules.
Furthermore, the melting behavior is affected by particle size and specific surface area. In this study, the raw materials exhibited grain sizes typical of casting powders. The same batches were used throughout all investigations, and identical treatments were applied to each mixture. Consequently, it is assumed that the melting behavior primarily depends on the raw materials employed, which may vary in grain size, similar to standard spray-dried products. Nevertheless, the grain size distributions of the respective raw materials must be optimized to meet process requirements. Such optimization is particularly necessary when selecting samples for application in the continuous casting process.
Comparisons of X-ray microanalysis of the homogeneous slag obtained after liquefaction of the investigated samples with the chemical composition given in Table 1 are in good agreement. This indicates the formation of a uniform mold slag layer below 1200°C in the continuous casting process without changing flow and crystallization behavior.
The results revealed in this study suggest the application of a mold powder containing no melt-controlling additive for steels sensitive to re-carburization. Nevertheless, it must be considered that service conditions cannot be represented in laboratory experiments. Thus, in the next step, the applicability of this mold powders has to be tested in the continuous casting process. The mold powder could be produced similar to the procedure suggested in this paper, with minor modifications for industrial scale. During spray-drying, difficulties may arise when processing basic granules with a high soda content because it exceeds the solubility limit of the slurry. Furthermore, a delay in the formation of new phases affecting the melting behavior can be expected, resulting from reduced contact between particles within a granule caused due to the formation of hollow spheres. Therefore, further investigation is required.
Funding
The authors gratefully acknowledge the funding support of K1-MET GmbH, a metallurgical competence center. The research program of the K1-MET competence center is supported by COMET (Competence Center for Excellent Technologies), the Austrian program for competence centers (grant number 892415). COMET is funded by the Federal Ministry for Climate Action, Environment, Energy, Mobility, Innovation and Technology, the Federal Ministry for Digital and Economic Affairs; the Federal States of Upper Austria, Tyrol, and Styria; and the Styrian Business Promotion Agency (SFG). Furthermore, thanks go to Upper Austrian Research GmbH for their continuous support. In addition to public funding from COMET, partial financing comes from the industrial partners RHI Magnesita, voestalpine Stahl, and voestalpine Stahl Donawitz, and the scientific partner Technical University Leoben.
Conflicts of interest
The authors have nothing to disclose.
Data availability statement
Data associated with this article cannot be disclosed due to legal reason.
Author contribution statement
Conceptualization, N. Gruber and H. Harmuth; Methodology, N. Gruber; Validation, N. Gruber and H. Harmuth; Formal Analysis, N. Gruber and H. Harmuth; Investigation, N. Gruber; Resources, N. Gruber; Data Curation, N. Gruber; Writing – original draft Preparation, N. Gruber; Writing – review and editing, H. Harmuth.; Visualization, N. Gruber and H. Harmuth; Supervision, H. Harmuth; Project administration, N. Gruber and H. Harmuth; Funding acquisition, N. Gruber and H. Harmuth.
References
- K.C. Mills, A.B. Fox, R.P. Thackray, et al., The performance and properties of mould fluxes, Proc. of VII Int. Conf. on Molten Slags, Fluxes & Salts, The South African Institute of Mining and Metallurgy, Johannesburg. 713 (2004) [Google Scholar]
- K.C. Mills: NPL Rep. DMM(A), A review of ECSC funded research on mould powders during continuous casting, ECSC Contract No. 7210.22/451, Report EUR 13177 EN, Teddington, U.K. (1990) [Google Scholar]
- M. Kawamoto, K. Nakajima, T. Kanazawa, K. Nakai, Design principles of mold powder for high speed continuous casting, ISIJ Int. 34, 593 (1994) [Google Scholar]
- N. Kölbl, I. Marschall, H. Harmuth, Investigation of the melting behaviour of mould powders, in: M. Sanchez: Proc. of VIII Int. Conf. on Molten Slags, Fluxes & Salts, Santiago, Chile. 1031 (2009) [Google Scholar]
- J.A. Kromhout, Mould powders for high speed continuous casting of steel, PhD Thesis, Technical University Delft. 2013. http://resolver.tudelft.nl/uuid:4b52997d-9fc9-41b3-a4e8-70ffab1b3fdf. Accessed 05 April 2023 [Google Scholar]
- M. Supradist, A. Cramb, K. Schwerdtfeger, Combustion of carbon in casting powder in a temperature gradient, ISIJ Int. 44, 817 (2004) [Google Scholar]
- Z. Yi, J. Song, Q. Peng, Analysis of Carbonization mechanism in ULCS continuous casting and control measures, Adv. Mat. Res. 739, 214 (2013) [Google Scholar]
- Q. Long, W. Wang, K. Dou, Investigation on carbonizing from mold flux into ultra-low-carbon steel during continuous casting. Metall. Mater. Trans. 54B, 263 (2023) [Google Scholar]
- S. Terada, S. Kaneko, T. Ishikawa et al., Development of mold fluxes for ultra-low-carbon steels, Iron Steelmak. 18, 41 (1991) [Google Scholar]
- P. Cheng, O. Li, Z. Duan et al., Kinetics of MnO2-catalyzed combustion of carbonaceous materials in mold flux, Fuel. 397, 135411 (2025) [Google Scholar]
- F. Han, G. Wen, P. Tang et al., Catalytic effect of iron oxide on the combustion of carbonaceous materials in mold flux for continuous casting. Metall. Mater. Trans. B. 54, 2605 (2023) [Google Scholar]
- H. Nakato, S. Takeuchi, T. Fujii et al., Characteristics of new mold fluxes for strand casting of low and ultra-low carbon steel slabs, in: 75th Steelmak. Conf. Proc., Washington DC, USA. 639 (1991) [Google Scholar]
- H. Takeuchi, H. Mori, T. Nishida et al., Development of a carbon-free casting powder for continuous casting, Tetsu-to-Hagané, 64, 1548 (1978) [Google Scholar]
- H. Takeuchi, T. Nishida, T. Ohno et al., Kohlenstofffreies gießpulver für kokillen- und strangguß von stahl. Deutsches Patent 26, 354 (1976) München, Deutschland: Deutsches Patentamt. [Google Scholar]
- B. Debiesme, J.-P. Radot, D. Coulombet et al., Mold cover powder for continuous casting of steel, especially very-low-carbon steels, United States Patent (5,876,482), (1996) Washington D.C.: U.S. Patent and Trademark Office [Google Scholar]
- B. Debiesme, J.-P. Radot, D. Coulombet et al., Mold cover powder for continuous casting of steel, especially very-low-carbon steels, United States Patent (6,328,781), (1998) Washington D.C.: U.S. Patent and Trademark Office. [Google Scholar]
- C. Lefebvre, J.P. Radot, J.N. Pontoir et al., Dévelopment d’une poudre de lingotière permettant de limiter la recarburation des aciers à ultra-ba carbone, La Revue de Métallurgie-CIT. 489 (1996) [Google Scholar]
- l. Sun Ch. Liu, J. Fang et al., Crystallization behavior and thermal properties of B2O3− containing MgO−Al2O3−SiO2−Li2O glass-ceramic and its wettability on Si3N4 ceramic, J. Eur. Ceram. Soc. 39, 1532 (2019) [Google Scholar]
- J.J. Macho, G. Hecko, B. Golinmowski et al., The development and evaluation of a new generation of no free carbon continuous casting fluxes, in: Preprints 33rd McMaster Symp. on Iron and Steelmaking, McMaster Univ., Hamilton, CN. 131 (2005) [Google Scholar]
- J. Safarian, M. Tangstad, Wettability of silicon carbide by CaO−SiO2 slags, Metall. Mat. Trans. 40B, 920 (2009) [Google Scholar]
- J. Park, J. Jeon, K. Lee et al., Initial wetting and spreading rates between SiC and CaO-SiO2-MnO Slag. Metall. Mater. Trans. 47B, 1832 (2016) [Google Scholar]
- O. Maillart, V. Chaumat, F. Hodaj, Wetting and interfacial interactions in the CaO−Al 2 O3−SiO2/silicon carbide system, J. Mater. Sci. 45, 2126 (2010) [Google Scholar]
- L. Ning, Ch. Jinghao, Silicon carbide as melting rate regulator of mould flux for continuous casting of ULC steels, J. Shaoguan University. 19, 112 (1998) [Google Scholar]
- Q.C. Peng, L. Deng, J. L Song et al., Carbonization Research in ULCS Continuous Casting, AMR. 1089, 315 (2015) [Google Scholar]
- M.-S. Park, S. Yoo, Effect of carbon pickup on the slab with slag pool thickness in ultra-low carbon steel, in: R. G. Reddy, P. Chaubal, P. Ch. Pistorius and U. Pal: Proc. 10th Int. Conf. on Molten Slags, Fluxes and Salts, TMS (The Minerals, Metals & Materials Society), 343 (2016) [Google Scholar]
- M. Radu, T. Heput, E. Ardelean, A. Dascal, Research on the influence of casting powders on the higher quality of continuous cast semifinished parts, J. Phys. Conf. Ser. 1781, 012057 (2021) [Google Scholar]
- T. Zhang, H. Long, G. Cheng et al., Cause of bending fracture in high-strength fasteners and heredity of surface recarburization in high carbon steel, Engineering Failure Analysis. 160, 1350 (2024) [Google Scholar]
- N. Gruber, Liquefaction controlling components and their effect on carbon-free mold powders, Metall. Mat. Trans. 54B, 3092 (2023) [Google Scholar]
- N. Gruber, Effect of liquefaction controlling components in carbon-free mould powder for the continuous casting of ultra-low carbon steels, in: P. Hayes and E. Jak: Proc. 12th Int. Conf. on Molten Slags, Fluxes and Salts, The Australian Institute of Mining and Metallurgy. 979 (2024) [Google Scholar]
- Taikabutsu Gijutsu Kyōkai (Japan): Refractories handbook, the technical association of refractories, Japan. 1998 [Google Scholar]
- T. Wang, A. Yamaguchi, Oxidation Protection of MgO−C Refractories by Means of Al8B4 C 7, J. Am. Ceram. Soc. 84, 577 (2001) [Google Scholar]
- N.K. Ghosh, K. P. Jagannathan, D.N. Ghosh, Oxidation of magnesia-carbon refractories with addition of aluminum and silicon in air, Interceram. 50, 196 (2001) [Google Scholar]
- L. Liu, X. Han, D. Zhang et al., Effects of mineral raw materials on melting and crystalline properties of mold flux and mineralogical structures of flux film for casting peritectic steel slab, ISIJ Int. 63, 1697 (2023) [Google Scholar]
- I. Marschall, N. Kölbl, H. Harmuth et al., Identification of secondary raw materials in mold powders and their melting behavior, J. Iron Steel Res. Int. 26, 403 (2019) [Google Scholar]
- F. Han, L. Yu, G. Wen et al., The effect of composition segregation of mold powder produced by spray granulation on the sintering performance, J. Mat. Res. Techn. 20, 448 (2022) [Google Scholar]
Cite this article as: Nathalie Gruber, Harald Harmuth, Concepts for the development of carbon-free mold powders for the continuous casting of steels, Metall. Res. Technol. 123, 211 (2026), https://doi.org/10.1051/metal/2025152
All Tables
Mold slag composition after total liquefaction of MP 0 in wt% (The fluorine content is expressed once in elemental form and once in the form of CaF2).
All Figures
 |
Fig. 1 Backscattered electron image of (a) a basic and (b) an acidic granule of sample 1 after annealing at 1000°C: 1 fluorite, 2 wollastonite, 3 SiC, 4 cuspidine, 5 residuum of magnesite, 6 magnesium fluoride, 7 fluorine-containing liquid phase, 8 albite. |
| In the text | |
 |
Fig. 2 Backscattered electron image of (a) a basic and (b) an acidic granule of sample MP 3 after annealing at 1000°C: 1 magnesium fluoride, 2 calcium magnesium fluoride, 3 cuspidine, 4 liquid phase, 5 fluorite, 6 wollastonite, 7 SiC, 8 albite. |
| In the text | |
 |
Fig. 3 Backscattered electron image of (a) MP 4 and (b) MP 5 after annealing at 1000°C: 1 cuspidine, 2 wollastonite, 3 quartz, 4 hematite, 5 fluorite, 6 diopside, 7 Ca2SiO4, 8 calcite, 9 magnesite, 10 calcium magnesium fluoride, 11 magnesium fluoride, 12 liquid phase. |
| In the text | |
 |
Fig. 4 Backscattered electron image of sample MP 4 after annealing at 1100°C: 1 cuspidine, 2 wollastonite, 3 quartz, 4 feldspar, 5 fluorite, 6 diopside, 7 liquid phase, 8 calcium magnesium fluoride, 12 liquid phase. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


